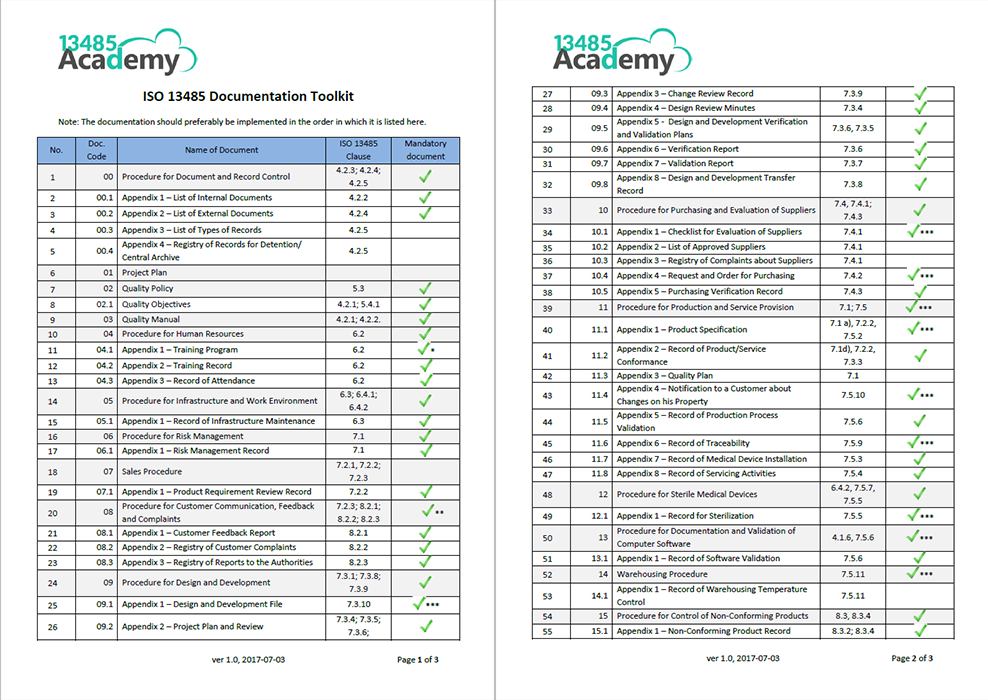 
Certificates are issued by a third-party registrar following a certification audit, and everyone in your organization will need to be involved in the process. You must complete several steps to achieve and maintain certification. With an ISO 13485 management system, you get more value from your operations, increasing productivity and cost-efficiency across your organization. ISO 13485 certification also streamlines your day-to-day manufacturing processes. They prove your commitment to quality and safety, building trust with consumers as you continually deliver safe, effective products and components. The medical device manufacturing requirements of ISO 13485 set your business apart. With ISO-compliant quality management, you can reduce risk in the supply chain and increase customer satisfaction.īy meeting ISO 13485 requirements, you empower your business to move forward and engage new markets. The International Organization for Standardization (ISO) developed ISO 13485 to standardize the quality and safety requirements for medical devices worldwide. In the medical device industry, quality and safety are crucial.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
